- 1010 cm⁻¹ (height 0.71, strong sharp): Likely C–O stretch (alkoxy or ether). Standard range: 1000–1200 cm⁻¹ for C–O single bonds. Sharp, strong peak suggests a well-defined ether or alcohol group. - 1017 cm⁻¹ (height 1.00, strong sharp): Also consistent with C–O stretch, possibly from a secondary alcohol or ether. The higher intensity suggests a dominant or symmetric C–O bond. - 3268 cm⁻¹ (height 0.24, weak broad): O–H stretch (alcohol or carboxylic acid). Broad, weak peak indicates hydrogen bonding, typical of alcohols or weakly acidic O–H. - 3282 cm⁻¹ (height 0.24, weak broad): Also O–H stretch, possibly from a different hydrogen-bonded environment (e.g., intermolecular H-bonding). - 3308 cm⁻¹ (height 0.24, weak broad): Another O–H stretch, consistent with broad, weak signals from hydrogen-bonded hydroxyl groups.
Conclusion: The spectrum suggests a compound containing multiple hydroxyl groups (alcohol or phenol) and ether linkages. The strong, sharp C–O peaks at 1010 and 1017 cm⁻¹ indicate ether or alcohol functional groups, while the weak, broad O–H peaks at 3268–3308 cm⁻¹ confirm the presence of hydrogen-bonded hydroxyls. The absence of a sharp C=O peak (~1700 cm⁻¹) rules out carboxylic acids or ketones. The sample is likely an alcohol or polyol (e.g., a sugar or polyether alcohol) with multiple –OH and C–O bonds.
This discussion presents an infrared spectral analysis combining
automated interpretation with reference comparison to support
functional group identification and structural assessment.
FTIR Spectrum Interpretation Summary
Comparative Analysis Conclusion
AI-assisted Interpretation Conclusion
- 1010 cm⁻¹ (height 0.71, strong sharp): Likely C–O stretch (alkoxy or ether). Standard range: 1000–1200 cm⁻¹ for C–O single bonds. Sharp, strong peak suggests a well-defined ether or alcohol group.
- 1017 cm⁻¹ (height 1.00, strong sharp): Also consistent with C–O stretch, possibly from a secondary alcohol or ether. The higher intensity suggests a dominant or symmetric C–O bond.
- 3268 cm⁻¹ (height 0.24, weak broad): O–H stretch (alcohol or carboxylic acid). Broad, weak peak indicates hydrogen bonding, typical of alcohols or weakly acidic O–H.
- 3282 cm⁻¹ (height 0.24, weak broad): Also O–H stretch, possibly from a different hydrogen-bonded environment (e.g., intermolecular H-bonding).
- 3308 cm⁻¹ (height 0.24, weak broad): Another O–H stretch, consistent with broad, weak signals from hydrogen-bonded hydroxyl groups.
Conclusion: The spectrum suggests a compound containing multiple hydroxyl groups (alcohol or phenol) and ether linkages. The strong, sharp C–O peaks at 1010 and 1017 cm⁻¹ indicate ether or alcohol functional groups, while the weak, broad O–H peaks at 3268–3308 cm⁻¹ confirm the presence of hydrogen-bonded hydroxyls. The absence of a sharp C=O peak (~1700 cm⁻¹) rules out carboxylic acids or ketones. The sample is likely an alcohol or polyol (e.g., a sugar or polyether alcohol) with multiple –OH and C–O bonds.
This discussion presents an infrared spectral analysis combining automated interpretation with reference comparison to support functional group identification and structural assessment.
raw image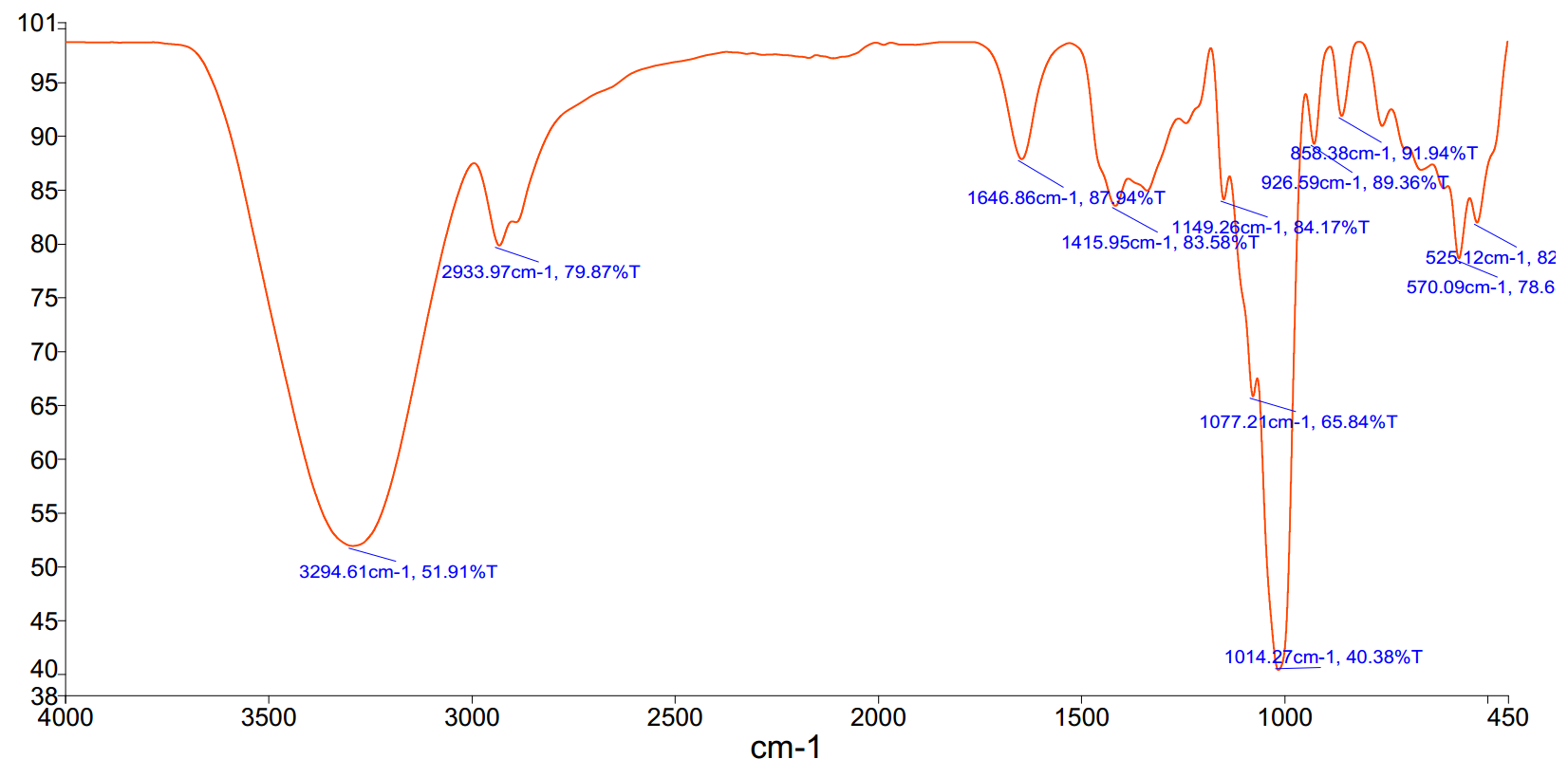 tem.csv
tem.csv
en&2