**Data:** * **770 cm⁻¹ (weak, sharp):** Out-of-plane C-H bending (aromatic, monosubstituted or 1,2,4-trisubstituted typical range: 770-730 cm⁻¹ & 710-690 cm⁻¹). Only one peak in this region is observed. * **826 cm⁻¹ (medium, broad):** C-O-C stretching (possibly from epoxide or cyclic ether, typical range: ~1250-800 cm⁻¹, often broad). Could also be C-Cl stretching. * **924, 936, 944 cm⁻¹ (weak/medium, sharp):** =C-H out-of-plane bending (terminal alkene, ~990 & 910 cm⁻¹) or C-O stretching (various). * **1029 cm⁻¹ (medium, broad):** C-O stretching (primary alcohol, ether, or epoxy group, typical range: ~1100-1000 cm⁻¹). * **1178, 1194 cm⁻¹ (medium, sharp):** C-O stretching (ester, ether, or phenolic C-O, typical range: ~1300-1000 cm⁻¹) or C-C skeletal vibrations. * **1246 cm⁻¹ (medium, broad):** C-O-C asymmetric stretching (aryl alkyl ether, typical ~1250 cm⁻¹) or C-O stretching (phenolic compound). * **1293 cm⁻¹ (weak, sharp):** C-O stretching or C-H bending. * **1366 cm⁻¹ (weak, sharp):** Symmetric CH₃ bending (gem-dimethyl, typical ~1370 cm⁻¹) or O-H bending (phenolic). * **1503, 1511, 1519 cm⁻¹ (strong, sharp):** Aromatic C=C ring stretching (typical range: ~1600-1450 cm⁻¹). The triplet suggests a substituted benzene ring, possibly with electron-withdrawing groups. * **1621 cm⁻¹ (weak, broad):** Conjugated C=C stretching (alkene or aromatic overtone/combination band, typical ~1600-1580 cm⁻¹) or adsorbed water. * **2933, 2945, 2973, 2981 cm⁻¹ (weak, broad):** Aliphatic C-H asymmetric stretching (CH₃, CH₂, typical range: ~2975-2850 cm⁻¹). The weak intensity suggests low aliphatic content.
**Analysis:** 1. The strong, sharp triplet between 1503-1519 cm⁻¹ is definitive evidence for an **aromatic ring**. 2. The single weak peak at 770 cm⁻¹ suggests a specific, likely polysubstituted, aromatic substitution pattern (not monosubstituted, which typically shows two strong bands). 3. The cluster of medium-intensity, sharp/broad bands between 1246-826 cm⁻¹ is dominated by **C-O stretching vibrations**, indicating the presence of **ether (C-O-C), epoxy, or phenolic (Ar-O) groups**. The broadness of some peaks (826, 1029, 1246 cm⁻¹) is characteristic of C-O-C stretching. 4. The weak peaks between 924-944 cm⁻¹ could indicate trace alkene or further C-O vibrations. 5. The weak, broad C-H stretches above 2900 cm⁻¹ indicate minor aliphatic (CH₃/CH₂) content. 6. The weak, broad peak at 1621 cm⁻¹ may be a conjugated system component or an artifact (e.g., water).
**Conclusion/Inference:** * **Primary Component:** The spectrum is dominated by an **aromatic compound containing significant C-O functionality**. * **Probable Functional Groups Present:** * **Aromatic ring** (confirmed, likely polysubstituted). * **Ether (C-O-C) and/or phenolic (Ar-OH) groups** (strongly indicated by the C-O stretching region profile). * Minor **aliphatic (CH₃/CH₂) groups**. * **Inference on Compound Class:** Based on the pattern (strong aromatic bands, prominent C-O-C/C-O stretches, weak aliphatic stretches), the sample is most likely a **polysubstituted aromatic ether, a phenolic resin derivative, or an epoxy-aromatic compound**. The absence of a strong, broad O-H stretch (~3400-3200 cm⁻¹) suggests the phenolic O-H may be substituted (e.g., in an ether or epoxy) or the phenol concentration is low. The specific substitution pattern cannot be determined from the provided data. * **Note:** The weak peaks in the 900-950 cm⁻¹ region remain ambiguous without further data; they could be part of the epoxy/ether system or indicate a trace component.
This discussion presents an infrared spectral analysis combining
automated interpretation with reference comparison to support
functional group identification and structural assessment.
FTIR Spectrum Interpretation Summary
Comparative Analysis Conclusion
AI-assisted Interpretation Conclusion
**Data → Analysis → Conclusion/Inference**
**Data:**
* **770 cm⁻¹ (weak, sharp):** Out-of-plane C-H bending (aromatic, monosubstituted or 1,2,4-trisubstituted typical range: 770-730 cm⁻¹ & 710-690 cm⁻¹). Only one peak in this region is observed.
* **826 cm⁻¹ (medium, broad):** C-O-C stretching (possibly from epoxide or cyclic ether, typical range: ~1250-800 cm⁻¹, often broad). Could also be C-Cl stretching.
* **924, 936, 944 cm⁻¹ (weak/medium, sharp):** =C-H out-of-plane bending (terminal alkene, ~990 & 910 cm⁻¹) or C-O stretching (various).
* **1029 cm⁻¹ (medium, broad):** C-O stretching (primary alcohol, ether, or epoxy group, typical range: ~1100-1000 cm⁻¹).
* **1178, 1194 cm⁻¹ (medium, sharp):** C-O stretching (ester, ether, or phenolic C-O, typical range: ~1300-1000 cm⁻¹) or C-C skeletal vibrations.
* **1246 cm⁻¹ (medium, broad):** C-O-C asymmetric stretching (aryl alkyl ether, typical ~1250 cm⁻¹) or C-O stretching (phenolic compound).
* **1293 cm⁻¹ (weak, sharp):** C-O stretching or C-H bending.
* **1366 cm⁻¹ (weak, sharp):** Symmetric CH₃ bending (gem-dimethyl, typical ~1370 cm⁻¹) or O-H bending (phenolic).
* **1503, 1511, 1519 cm⁻¹ (strong, sharp):** Aromatic C=C ring stretching (typical range: ~1600-1450 cm⁻¹). The triplet suggests a substituted benzene ring, possibly with electron-withdrawing groups.
* **1621 cm⁻¹ (weak, broad):** Conjugated C=C stretching (alkene or aromatic overtone/combination band, typical ~1600-1580 cm⁻¹) or adsorbed water.
* **2933, 2945, 2973, 2981 cm⁻¹ (weak, broad):** Aliphatic C-H asymmetric stretching (CH₃, CH₂, typical range: ~2975-2850 cm⁻¹). The weak intensity suggests low aliphatic content.
**Analysis:**
1. The strong, sharp triplet between 1503-1519 cm⁻¹ is definitive evidence for an **aromatic ring**.
2. The single weak peak at 770 cm⁻¹ suggests a specific, likely polysubstituted, aromatic substitution pattern (not monosubstituted, which typically shows two strong bands).
3. The cluster of medium-intensity, sharp/broad bands between 1246-826 cm⁻¹ is dominated by **C-O stretching vibrations**, indicating the presence of **ether (C-O-C), epoxy, or phenolic (Ar-O) groups**. The broadness of some peaks (826, 1029, 1246 cm⁻¹) is characteristic of C-O-C stretching.
4. The weak peaks between 924-944 cm⁻¹ could indicate trace alkene or further C-O vibrations.
5. The weak, broad C-H stretches above 2900 cm⁻¹ indicate minor aliphatic (CH₃/CH₂) content.
6. The weak, broad peak at 1621 cm⁻¹ may be a conjugated system component or an artifact (e.g., water).
**Conclusion/Inference:**
* **Primary Component:** The spectrum is dominated by an **aromatic compound containing significant C-O functionality**.
* **Probable Functional Groups Present:**
* **Aromatic ring** (confirmed, likely polysubstituted).
* **Ether (C-O-C) and/or phenolic (Ar-OH) groups** (strongly indicated by the C-O stretching region profile).
* Minor **aliphatic (CH₃/CH₂) groups**.
* **Inference on Compound Class:** Based on the pattern (strong aromatic bands, prominent C-O-C/C-O stretches, weak aliphatic stretches), the sample is most likely a **polysubstituted aromatic ether, a phenolic resin derivative, or an epoxy-aromatic compound**. The absence of a strong, broad O-H stretch (~3400-3200 cm⁻¹) suggests the phenolic O-H may be substituted (e.g., in an ether or epoxy) or the phenol concentration is low. The specific substitution pattern cannot be determined from the provided data.
* **Note:** The weak peaks in the 900-950 cm⁻¹ region remain ambiguous without further data; they could be part of the epoxy/ether system or indicate a trace component.
This discussion presents an infrared spectral analysis combining automated interpretation with reference comparison to support functional group identification and structural assessment.
RAW IMAGE: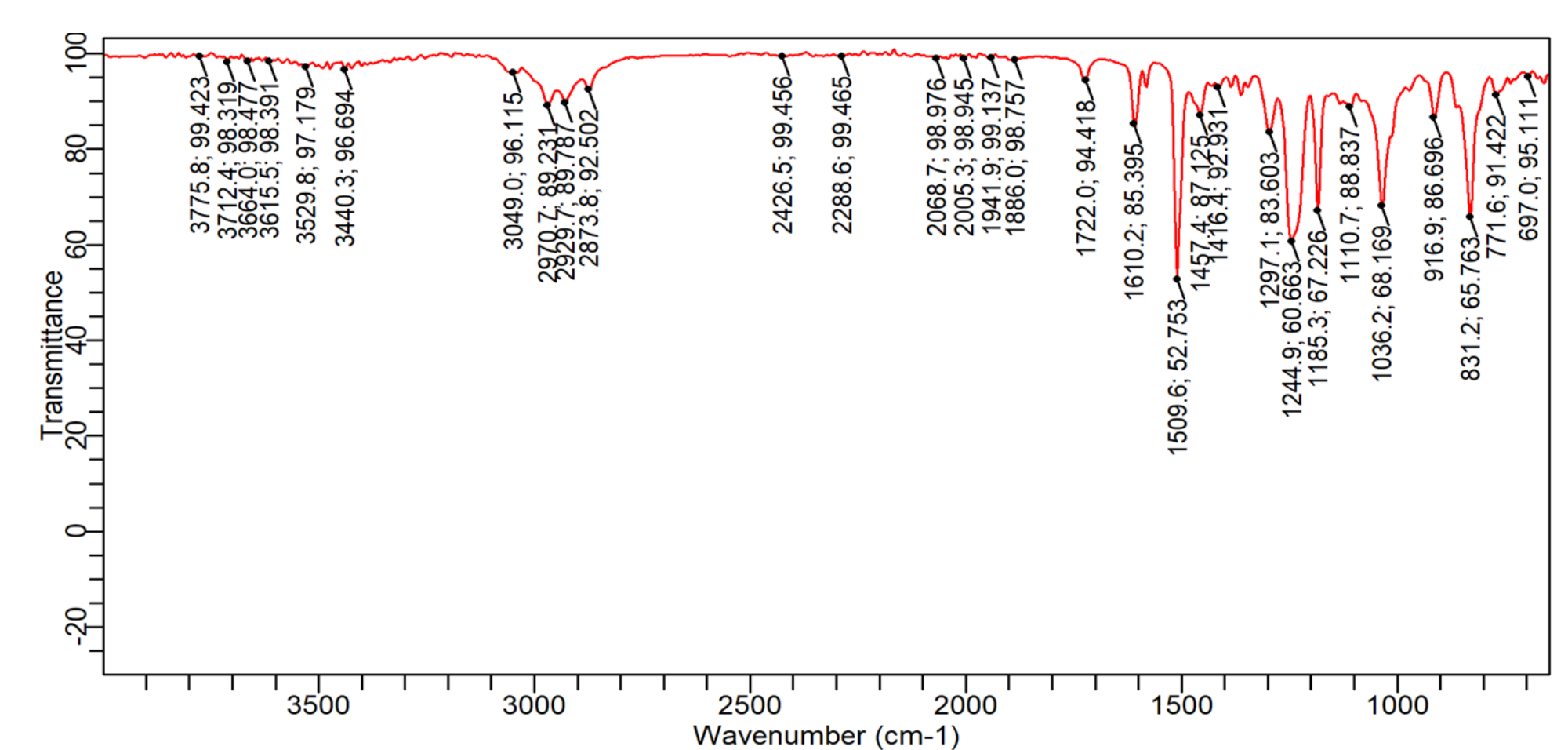
zh-hans&2